Home | Projects | CPGE | My carbon footprint
ZBRB bromine complexing agents (BCAs)
Classically in ZBRBs systems, quaternary ammonium bromides (QBr), such as N-ethyl-N-methyl-morpholinium bromide (MEM) and N-ethyl-N-methyl-pyrrolidinium bromide (MEP), are added the positive and negative half cell electrolytes.
CC[N+]1(C)CCOCC1.[Br-]
Fig. 1 : MEM
CC[N+]1(C)CCCC1.[Br-]
Fig. 2 : MEP
Experimentally, it has been shown that 0.75-0.8 M of total QBr can effectively eliminate bromine vapour at a temperature of 11°C, with all of the bromine effectively bound as an orange oily layer that settles to the bottom of the sample bottles. The orange oily layer seems to form only in the presence of both
Additionally, cyclic voltammetry (CV), linear sweep voltammetry and electrochemical impedance spectroscopy (EIS) studies also confirm that the addition of MEM/MEP effectively reduce
However, the addition of QBrs does result in a roughly 10% loss of cell efficiency.
Why is MEP predominantly used ?
Bromide ions reduced to bromine gas are stabilised in solution via a complexing agent, 1-ethyl-1-methylpyrrolidinium (MEP). As opposed to MEM, another complexing agent, dissolved MEP⁺ cations undergo a homogeneous chemical reaction with generated bromine. Thus, the storage as complex MEP-Br
Loss of cell efficency due to MEP-polybromide species
The complexation of poly-bromides by MEP⁺ forms either oil or solid phases, giving rise to new challenges. These phases tend to adhere to the membrane surface, blocking ion transport, and increasing the ohmic polarisation in the battery. Additionally, accumulated oil or solid phase acts as an insulation layer, reducing the number of active sites on the cathode and significantly damaging the battery performance.
To overcome these challenges, surfactants (e.g., polysorbate 20) can be added to the MEP-contained catholyte to disperse the bulk oil phase into small droplets, thereby easing phase separation at the electrolyte bottom. However, in general, the surfactant molecules are very large, reducing ion conduction, and are often unstable in an acidic mediums long term.
Source : 10.1002/adfm.202514730
Spectroscopic analysis
MEP-Br salt is colourless to pale, with no strong intrinsic visible absorption band.
However, molecular Br₂ has a visible absorption centered around 390 nm (3.18 eV), while Br₃⁻ has a strong UV band around 267-279 nm (4.59 eV). Therefore UV-vis spectrometry could be use to evaluate bromine concentration.
The first limitation is speciation. In water, bromine, bromide, and tribromide are in equilibrium:
This reaction reaches a theoretical constant equilibrium up to pH 6 : therefore
According to Beer Lambert's law :
The second limitation is the separation of aqueous and oily MEP-polybromide phases. UV-vis spectroscopy requires a homogeneous solution with no suspension, therefore surfactants cannot be used. In a recent 2025 nature energy article, researchers propose Br–SANa complexes as a substitute to MEP.
NS(=O)(=O)O[Na]
Pictured above, this complexing agent allows for a homogeneous solution, better suited to spectroscopic analysis.

However this limitation does not apply when studying the bromine shuttling across the separator, as the trace polybromide that diffuses to the anolyte side is not sufficient to precipitate into an oily phase.
Whats more, while UV-vis spectrometry could be used to determine absorbances at
Electrochemical analysis
Protocol : for this analysis a three electrode cell was set up (see below). 20 mL of test electrolyte was poured in the glass cell. A magnetic stirrer was used in the linear sweep voltammetry experiments for measurement of
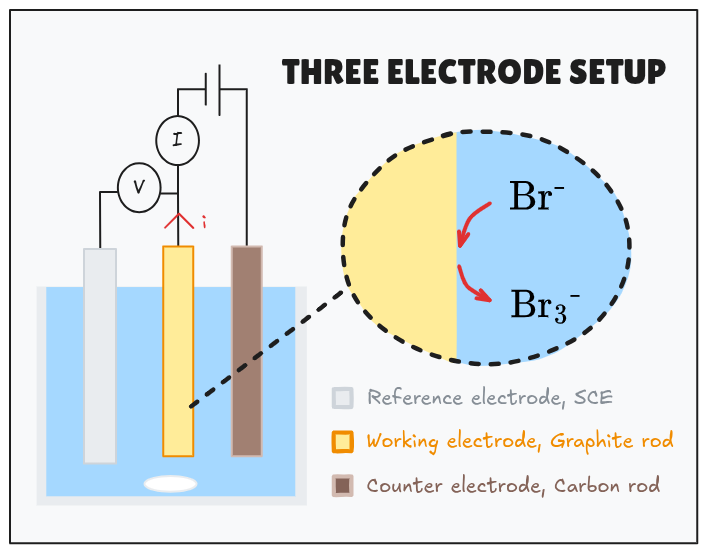
Source :
- Evaluation of N-ethyl-N-methyl-morpholinium bromide and N-ethyl-N-methyl-pyrrolidinium bromide as bromine complexing agents in vanadium bromide redox flow batteries http://dx.doi.org/10.1016/j.electacta.2013.06.084
Effective hydrodynamic/Stokes radius in water at ambient temperatures :
-
: : based on reported for aqueous triiodide. -
: : based on for tribromide in water. -
: no reliable tabulated aqueous hydrated radius in the literature, but polybromides are larger/slower than . Structurally, is also less sphere-like and can appear as V-shaped or chain-like motifs, so a single “radius” is less well defined.
Oil phase aqueous phase equilibrium
Due to the precipitation of