Home | Projects | CPGE | My carbon footprint
Zeta Potential
For colloidal solutions, one often speaks of the zeta potential (V), which characterizes the interfacial double layer (layer 1: Stern layer = condensed counter-ions, stationary, adsorbed on the particle; layer 2: slipping plane = mobile co-counter-ions)
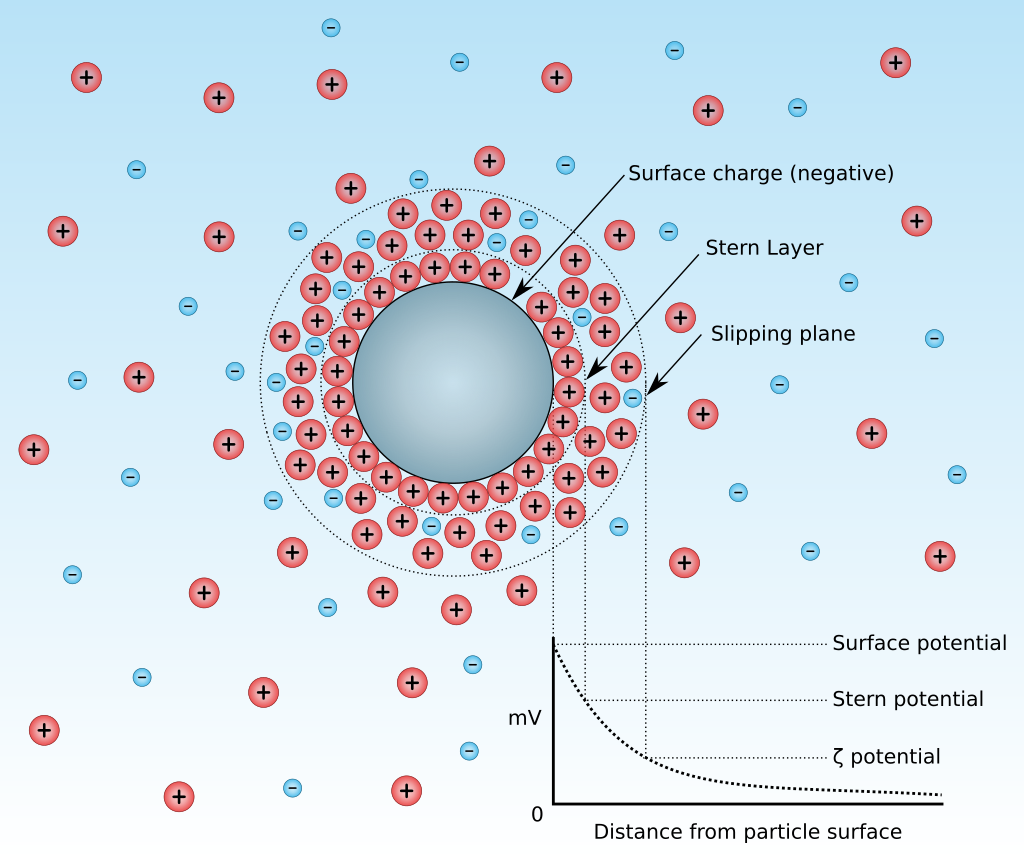
Key point: a high zeta potential
Point of zero charge (pzc)
The pzc is the pH at which the net electrical charge of the particle surface (e.g. the adsorbent's surface) is equal to zero. Generally, the pzc in electrochemistry is
From the view of the adsorbent, if the pH of the solution is below the pzc value, the surface charge of the adsorbent would become positive so that the anions can be adsorbed. Conversely, if the pH is above the pzc value, the surface charge would be negative so that the cations can be adsorbed.
Experimentally, curves are obtained of acid/base amount = f(pH), and pH=f(
Sources: