Home | Projects | CPGE | My carbon footprint
Electrolytes for chemical batteries
An electrolyte is a substance that conducts electricity through the movement of ions, but not through the movement of electrons : this includes most soluble salts, acids, and bases, dissolved in a polar solvent like water.
- Such a substance is called an ionic conductor
- The electrolyte is electrically neutral
- Gasses like HCl can also act as electrolytes, as well as molten salts
- Solid state batteries use solid ionic conductors as opposed to liquid electrolytes
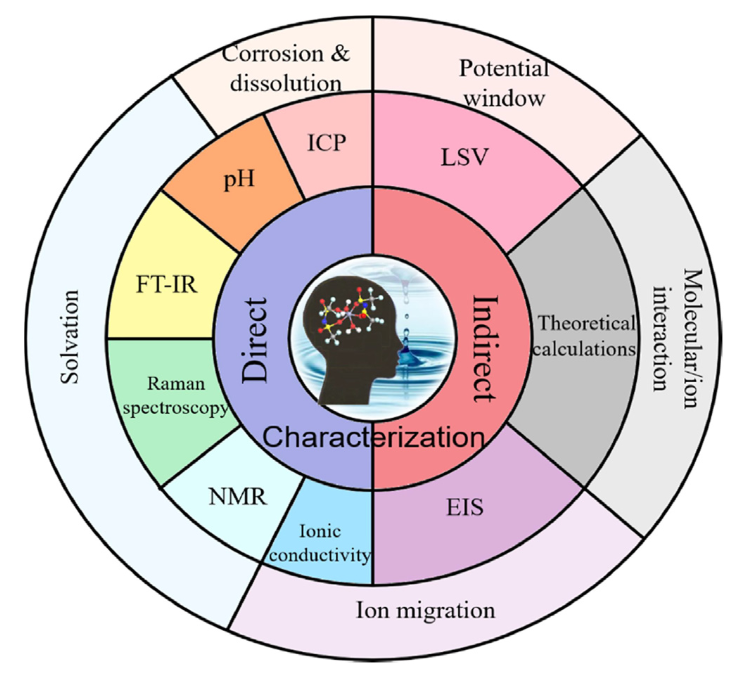
Characterisation of Electrolytes
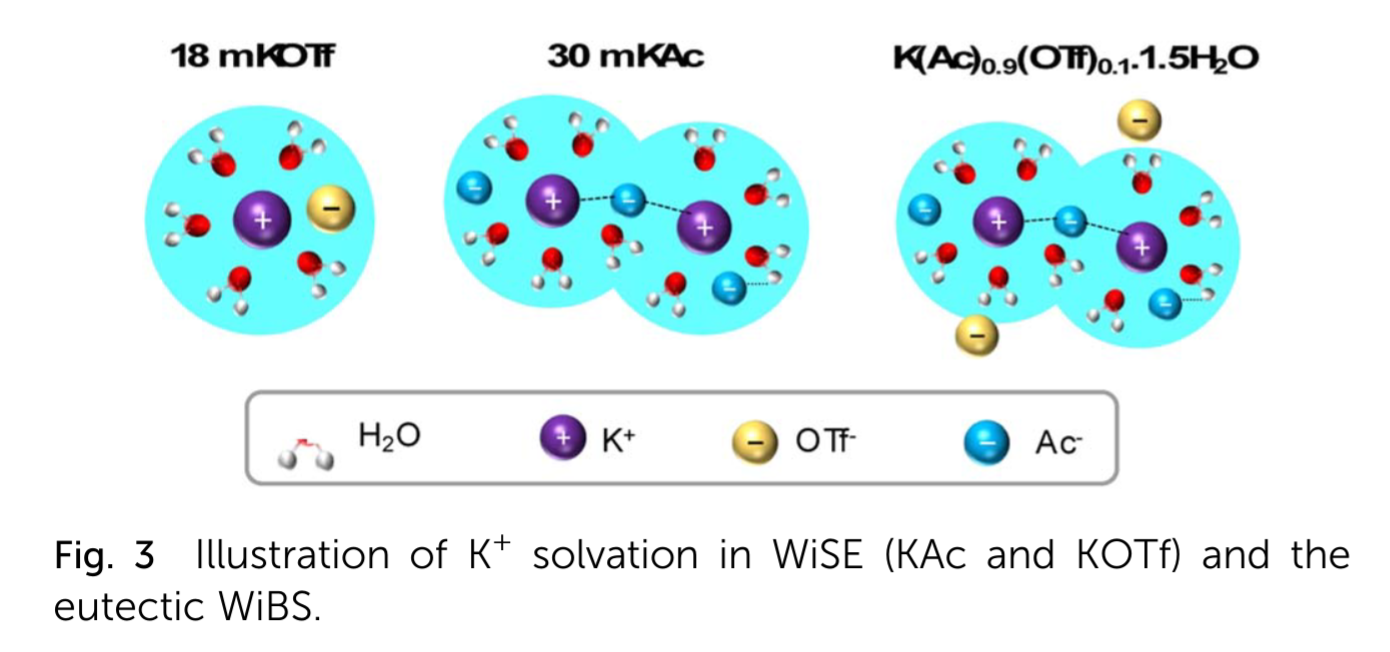
Water in Salt Electrolytes (WiSE) for aqueous batteries
WISEs are highly concentrated dissolves salts, having less water content than salt in terms of overall ratio. They may increase the electrochemical stability window (ESW) of aqueous batteries, as minority water content helps mitigate parasitic reactions like OER/HER. To achieve these conditions, various solvation sphere alteration approaches have been explored. Water can dissolve salt easily due to its high dielectric constant, but the minimal amount of water present in WiSEs means that these have a negligible tendency to dissolve electrode material.
An important phenomenon present in MIBs with organic electrolytes is the formation of the SEI, which protects the electrodes and enables a wider ESW. In WISEs there can be ionically conductive and electrically insulating passivation of the electrodes due to highly concentrated electrolyte composition. This helps prevent dendritic growth and also protects the electrode from corrosion by water by suppressing the water activity at its surface and thereby mitigating HER and OER.
- See the work of Jean-Marie Tarascon at the Collège de France
- Source : Water-in-Salt Electrolytes: Advances and Chemistry for Sustainable Aqueous Monovalent-Metal-Ion Batteries : https://doi.org/10.3390/batteries11040120
Further reading :
- For zinc ion electrolytes, see electrolytes for ZIBs
- For zinc bromine catholyte/anolyte, see electrolytes for ZBRBs