Home | Projects | CPGE | My carbon footprint
Aqueous electrolyte for Zinc Bromine Batteries
The operating environment for ZBRBs is relatively challenging: the electrolyte mainly consists of 1-2 M
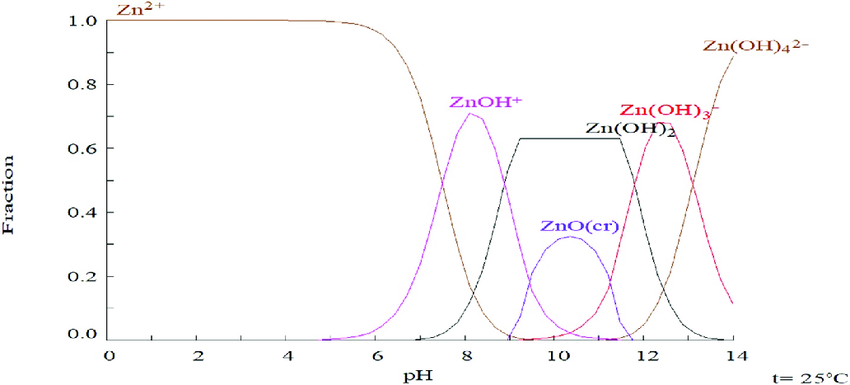
This low pH avoids formation of Zinc oxide/hydroxide species, as predicted by solubility-pH charts, that may passivate the anodic surface and decrease the capacity of the battery.
For bromine, at such acidic pH values, there is an equilibrium predicted between bromine gas and polybromide
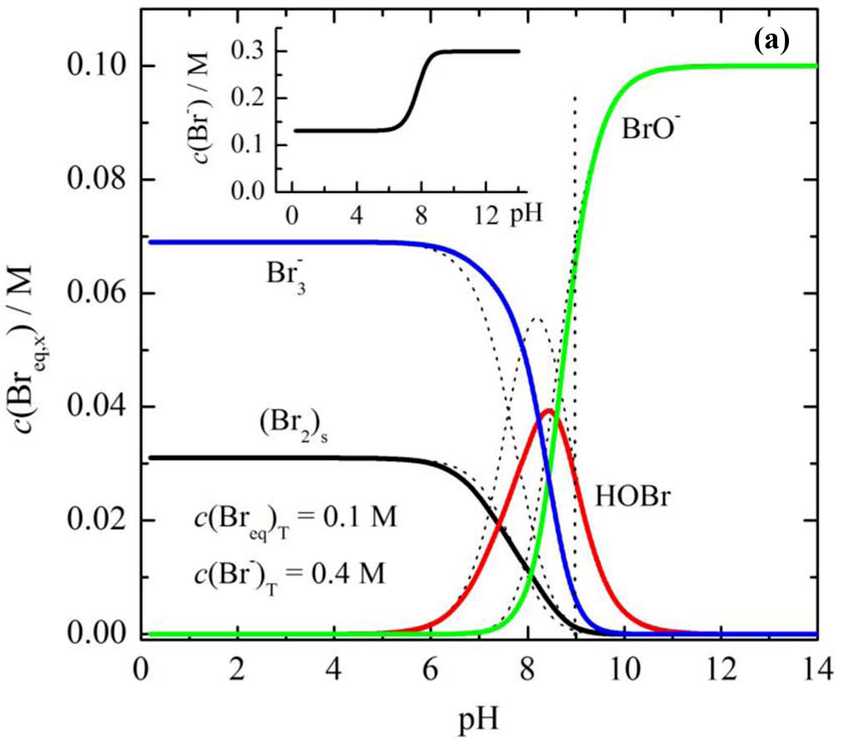
Image : Theoretical distribution of the equivalent bromine species as a function of pH for c(Br eq ) = 0.1 M, thin dash lines-for the constant c(Br -) = 0.131 M, thick full lines-for the bromine concentration shown in the inset
ZBRB electrolyte preparation protocol
In Bin Luo's lab the electrolyte used for ZBRBs is an aqueous solution of 2.0 M ZnBr₂ + 3.0 M KCl + 0.4 M MEP, and whilst their industry partner Redflow also added ZnCl₂ as a supporting salt, they do not.
Safety : zinc bromide is corrosive and toxic, and must be handled with proper PPE
For 100 mL of final electrolyte:
- Anhydrous ZnBr₂ at 99%: 2 M at 225.2 g/mol → 45.04 g
- KCl: 3 M at 74.55 g/mol → 7.46 g
- MEP bromine-complexing agent: 0.4 M at 202.2 g/mol → 8.089 g for 1-ethyl-1-methylpyrrolidinium bromide
Beware : these numbers are for the anhydrous salts in the lab. If new samples of ZnBr₂ or KCl are hydrated, the gram amounts need to be corrected to the hydrate’s molar mass.
Procedure :
- Add about 20 mL deionised water to a volumetric flask
- Add and dissolve ZnBr₂
- Add KCl and mix corked flask until clear
- Add the MEP bromine-complexing agent and mix
- Make up to 100 mL with deionised water, mix thoroughly leaving no bubbles
Notes :
- Add Zn salts gradually; these solutions can warm on dissolution.
- The volume of salts added is non negligible with regards to solution volume : be careful to not add too much water
- Use corrosion-resistant labware and good ventilation.
- Because bromine-complexing agents can change phase behaviour, mix until visually uniform.
Further reading :
- Another 2025 paper by Alghamdi et al. used a ZnBr₂ aqueous electrolyte with DMSO as a co-solvent additive, comparing 0% DMSO (control), 10% DMSO, and 30% DMSO.