Home | Projects | CPGE | My carbon footprint
Zinc-Bromine Rechargeable Batteries - Nano-Micro Letters (2023)
Glossary, safety and general information
- GF = Graphite Felt
- Anolyte : gives up
- Catholyte : accepts
- Membrane : lets
through - An aqueous solution of dissolved
1-4 M) is used as the electrolyte in most ZBRBs - Bromine smells like bleach, and
or polyhalide ions are highly oxidising - Bromine shuttle effect : diffusion of polybromide ions (resulting from
binding with ) from cathode to anode, leading to battery degradation (self discharge, capacity fade) - SoC = State of Charge (of the battery) : higher SoC increases overpotentials
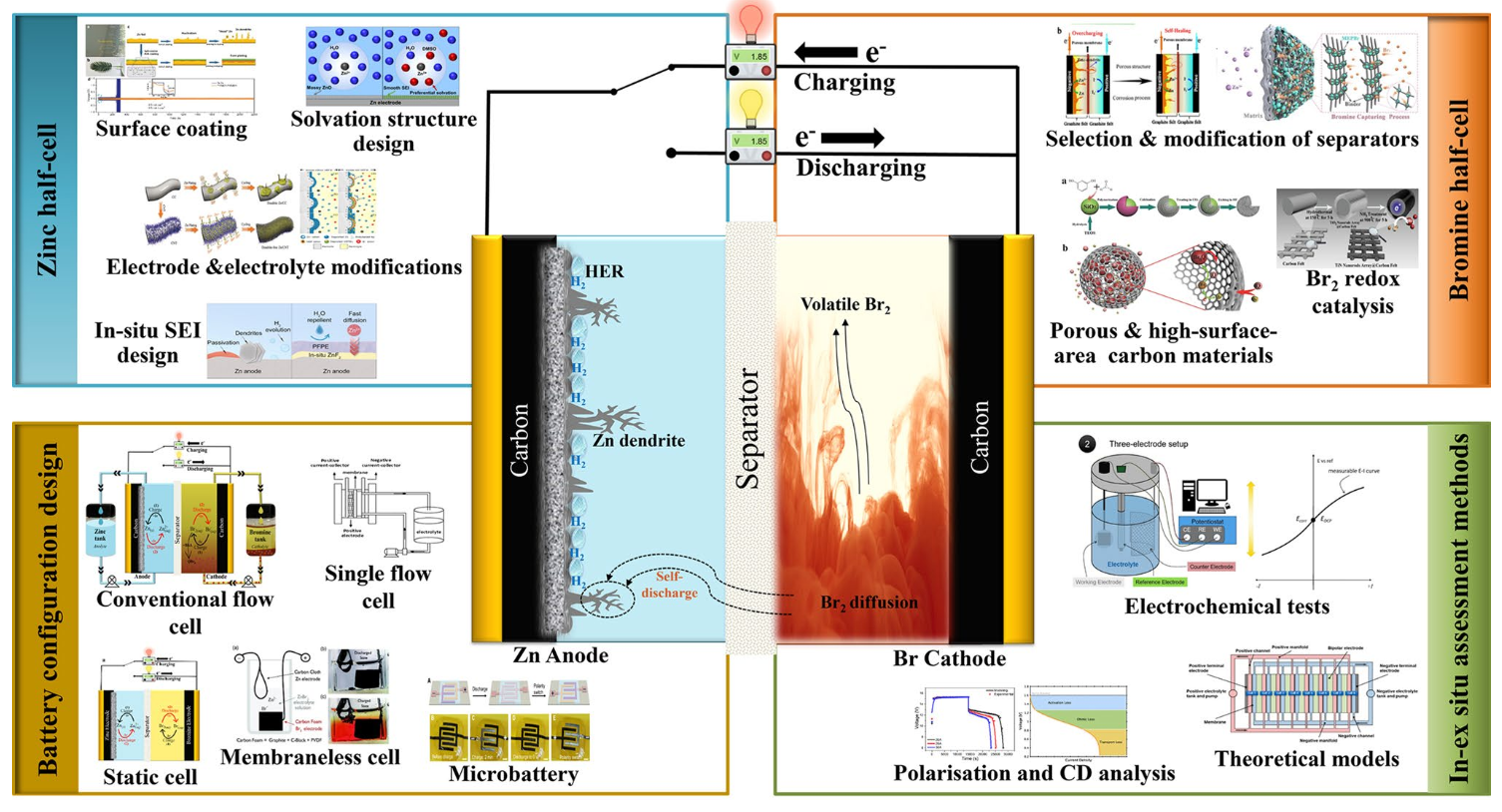
Zinc bromine rechargeable batteries (ZBRBs) are hybrid batteries : some of the energy is stored at the negative electrode (anode) via metallic zinc plated during the charging phase, while the remaining energy is stored in a liquid phase at the catholyte. They come in two configurations : static (cheaper, no pumping and lesser maintenance) and flow (higher efficiency). This study concentrates on flow ZBRBs.
Supporting Reactions
Anode side
+ 2e⁻ → (Charging) → + 2e⁻ (Discharging) - E°= −0.763 vs SHE at 25°C
Cathode side
- 2
→ + 2e⁻ (Charging) + 2e⁻ → 2 (Discharging) - E°=+1.065 V vs SHE at 25°C
Limitations of the technology
- Zinc dendrite growth resulting from repeated electroplating and stripping of zinc that can pierce the membrane and eventually forms a conductive bridge between the electrodes (shorting)
- hydrogen gas generation as the electrochemical potential of charge/discharge process of the system which is higher than that required for water hydrolysis which competes with the reduction reaction of Zn2+ ions and decreases the overall efficiency of the ZBRBs,
- corrosive elemental bromine liquid, Br 2 (l), production at the positive electrode during charge, which can be diffused through the membrane to the zinc half-cell reacting with the Zn plated at the negative electrode (crossover), causing self-discharge and/or degradation
- the low miscibility (~ 2.8 vol%) and stratification behaviour of Br2(l) in aqueous solutions that can lead to non-uniform concentration distribution
Static ZBRBs
- Held back by high self-discharge rate and low energy density (microbatteries suffer)
- glass fibre separators work well (instead of porous carbon)
- Gelion Endure™ company makes gel ZBRBs, but sensitive to temperatures over 50 °C
- Carbon foam electrode : highly porous flexible carbon foam
Redox Flow Battery (RFB)
- rapid response times, measured in milliseconds => these systems are well suited for levelling intermittent renewable power output
- VFRB (vanadium) are well-developed and commercialised
- ZBFB has substantial advantages over other flow batteries, such as high energy density, high cell voltage and the low cost of the materials
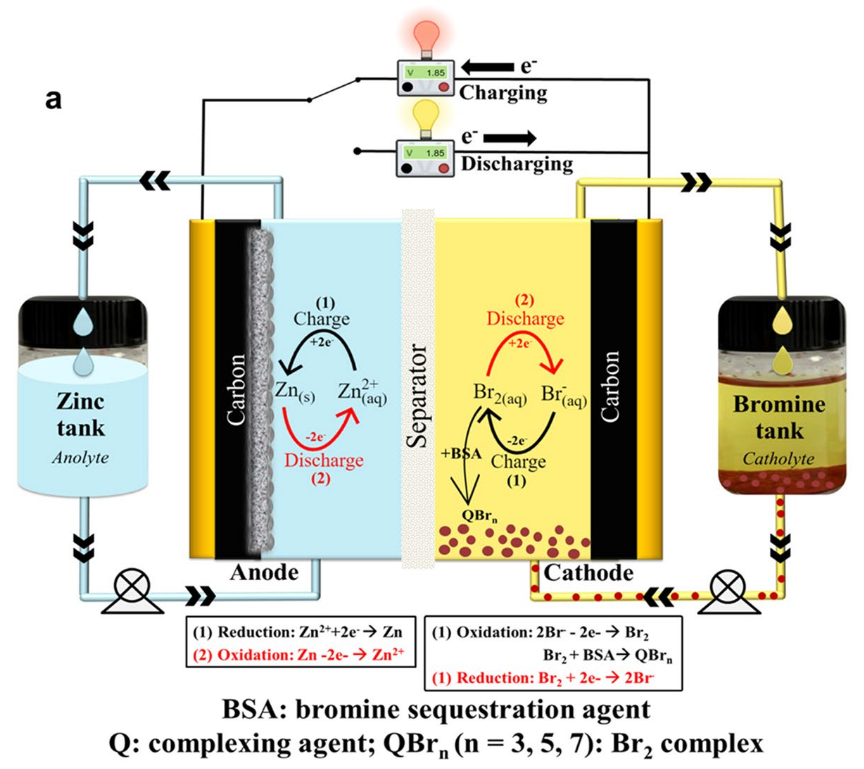
- Negative electrode (zinc), positive electrode (bromine), separated by a membrane to prevent cross-contamination.
- Two tanks of aqueous electrolyte solutions (anolyte and catholyte) contain electrochemically active species, including zinc (Zn2+) and bromide (Br-)
- Elemental bromine exists in equilibrium with bromide ions forming polybromide ions,
, where n = 3, 5 and 7 - two pumps to circulate the electrolyte solutions over both electrode surfaces, controlling generated heat, feeding and homogenising reactants, removing bromine complexes from the stack and ensuring uniform zinc deposits
- Single pump systems exist
Kinetics
- Behaviour and kinetics of zinc cations are strongly affected by other supporting electrolytes in aqueous solutions containing bromide
Dendritic growth (page 8)

- It is important to strip the zinc in ZBRBs for extended periods to ensure a smooth electrode surface for next zinc deposition.
- Residual zinc left on the anode after discharge results in the loss of 3–5% of the amp-hour capacity
- However, the remaining zinc could potentially be used as a useful energy source if additional zinc is plated over it in the subsequent cycles.
- @ What? Nucleation sites? Of course it can be stripped after, that doesn't make it additional energy
- Effect of operating temperature on zinc deposits : zinc deposits (grey in appearance) turned black at temperatures higher than 40 °C
- Smooth and bright zinc deposits were obtained when increasing the electrolyte’s zinc concentration
- In the initial stage, zinc deposition begins with nucleation and continues with growth, meaning the formation of dendrites is a cumulative result of battery cycling, not a single cycle.
- Zinc ions will deposit on a zinc nucleus rather than nucleating at a new site since zinc nucleation has a higher overpotential than zinc growth
- Formation of zinc dendrites can damage the membrane
- Dendrites form on the membrane side dominantly
Dendrite Mitigation Strategies
- Thermal treatment of GF => higher defect concentration => better zinc diffusion
- Aqueous electrolyte additives => reduced surface reactions
- Another proposed strategy involves creating an artificial interfacial layer between the zinc and the electrolyte that performs the same function as an SEI
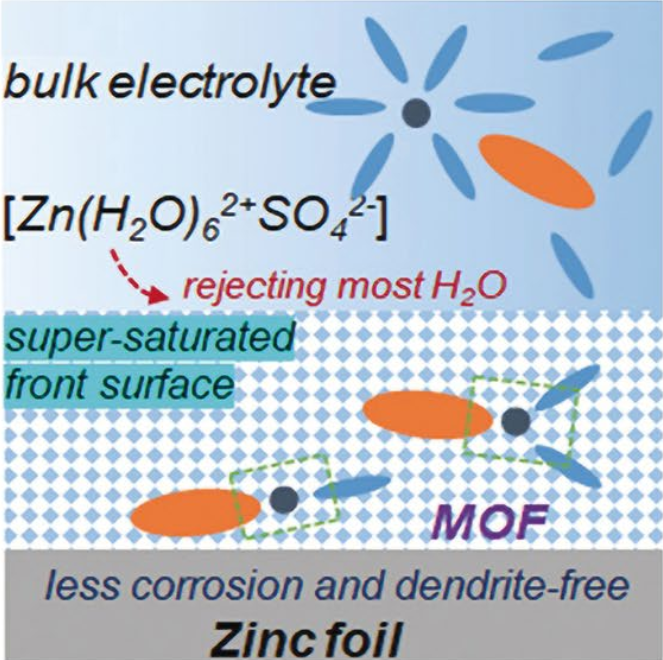
- Metal organic framework (MOF) material with unique channel structure can be used as coating layers to modify Zn anode through tuning specific chemical composition and porous structure. The ZIF-7 channel can prevent large solvated Zn ion complexes and create a supersaturated inner layer electrolyte to drive uniform metal deposition
- Organic additives (e.g. SDS) change surface morphology and surface orientation
- LiCl–ZnCl2 (water-in-salt) mixture-concentrated electrolyte on the hydrogen bonding interruption of water molecules and found that Zn2+ ions can coordinate with Cl− rather than H2O, leading to strong O–H covalent bonds while decreasing the solvation activity of H2O in the electrolyte
Bromine half-cell
In a Zn–Br flow battery, the fundamental positive-electrode electron-transfer reaction is usually written as :
- 2
→ + 2e⁻ (Charging) + 2e⁻ → 2 (Discharging)
However,
So the same cathode chemistry is often written in the overall form:
(Charging) (Discharging)
Conceptually, at the electrode bromide is oxidised to bromine. Then, in the electrolyte, bromine is redistributed into dissolved/complexed bromine species, especially
In practical batteries much of the bromine inventory is stored in these complexed forms rather than as “free”
Electrolyte
On top of
Membrane
- Must minimise the diffusion of
to the electroplated zinc, which causes self-discharge and lower CE for the system - In terms of morphology, membranes can be generally classified into porous and ion-exchange membranes (e.g. Nafion®), which are both appropriate and capable of separating the anode and cathode electrolytes in ZBFBs.
- Porous membranes are defined as macroporous (> 50 nm), mesoporous (2-50 nm) or microporous (0.2-2 nm) depending on their average pore diameter, while non-porous membranes transport ions via solution–diffusion mechanisms
Characterisation (page 22)
- Operando measurement = taken while a battery is operating (cycling)
- In situ measurement (meaning on site) = measuring a variable against a parameter relevant to the system, such as time, temperature, pressure or other variables, to minimise its degradation