Home | Projects | CPGE | My carbon footprint
Webinair : Electrode & Electrolyte Engineering in Aqueous Zinc-ion Batteries
Speaker : Professor Husam AlShareef
Why chose ZIBs ?
- Use case of ZIBs = low-cost, long life batteries for grid storage but not for high energy density applications like portable devices
Advantages of Zinc Batteries
- Much safer and more eco-friendly than lithium batteries
- Abundant and therefore cheap (bulk zinc costs $2/kg instead of $19/kg for lithium)
- Zinc is multivalent so there is higher potential charge density, as it has a similar ionic radius to lithium
- It has a suitable redox potential (-0.78V VS ESH) for use in aqueous electrolytes which are cheaper and less polluting
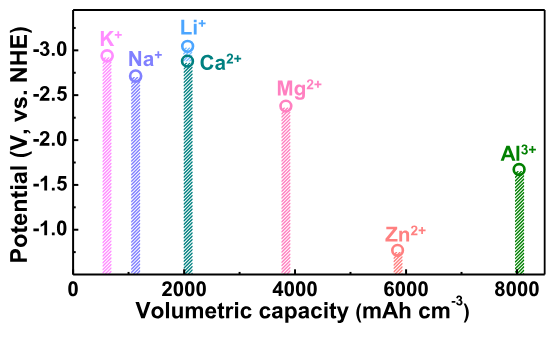
Fig. 1 : Volumetric capacity of different ions as a function of oxydation potential
History of Zinc Batteries
- 1882: Alkaline Zn-MnO2 single use batteries
- 1986 : Zn-MnO2ZnSO4 rechargeable battery
- 2010s : Stable zinc anodes
Issues with current ZIB technology
At the Cathode
- Dissolution
- Structural stability
- Surface by-products
At the Anode
- Zn corrosion (due to basic electrolytes)
- Surface by-products (ZHS at higher pH)
- Irreversible Hydrogen evolution reaction (
) - Dendrite formation (risking short circuiting or rupturing the cell)
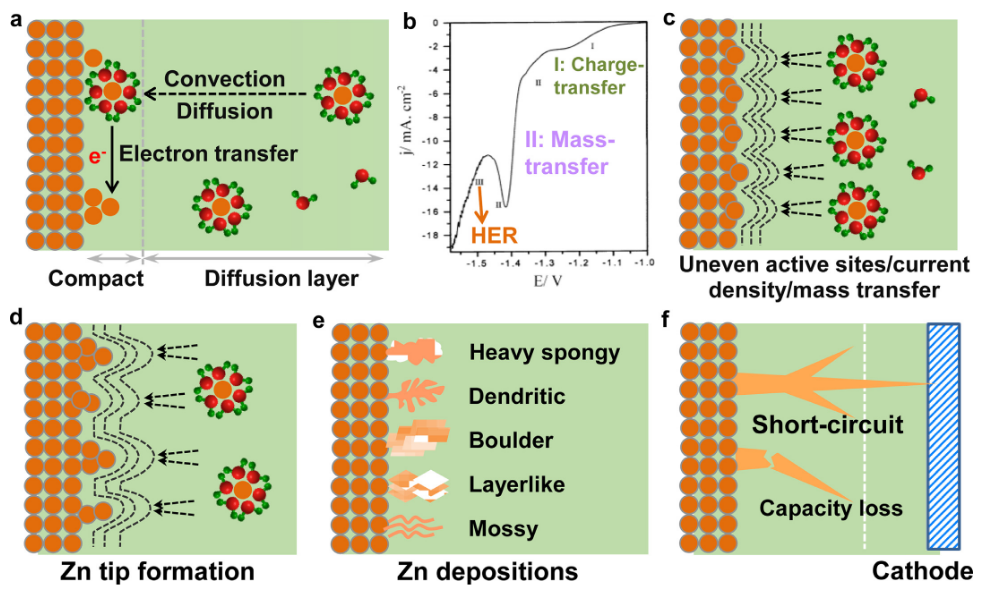
Fig. 2 : Dendrite formation mechanisms
With the Electrolyte
- Proton intercalation at cathode
- Electrolyte consumption at anode
- Undesired SEI chemistry
Chemical equations at play
There are multiple sets of chemical reactions at play, depending on pH and the imposed potential, as illustrated by the following figure :
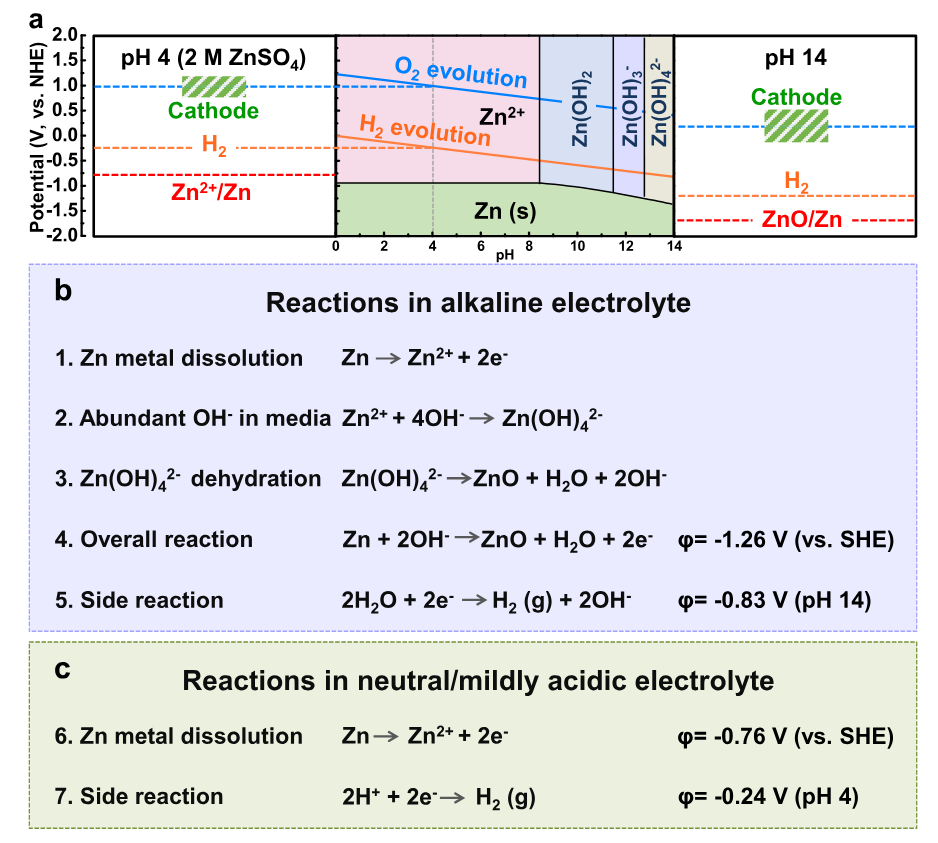
Fig. 3 : Working principle equations of ZIB batteries
HER and OER parasitic reactions
The evolution reactions of hydrogen at the cathode and oxygen at the anode are parasitic in nature, decreasing the overall efficiency and potentially damaging the battery cell. Their reaction equations are illustrated below :
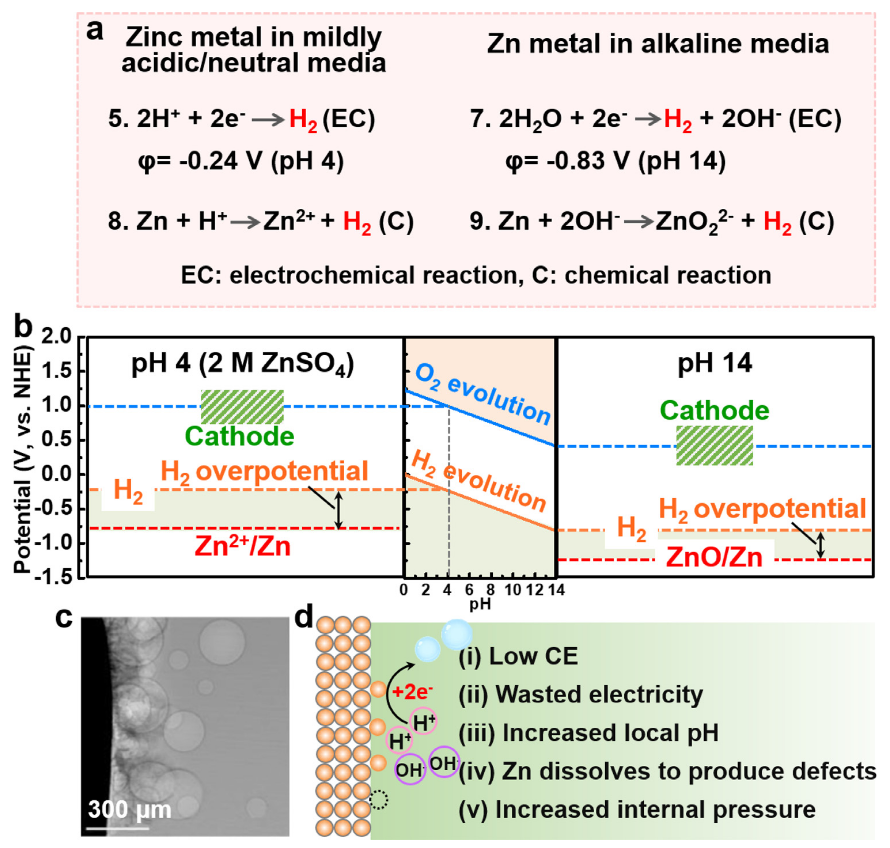
Fig. 4 : HER and OER reaction equations
Image Source: The rising zinc anodes for high-energy aqueous batteries, EnergyChem