Home | Projects | CPGE | My carbon footprint
MOF aSEIs for ZBRBs to avoid bromine diffusion and zinc dendrite growth
This project builds on a previous research project, Enhancing aqueous ZIBs by use of an artificial SEI of deposited ZIF-8 metal organic framework (MOF), but applied to Zinc Bromine Rechargeable Batteries (ZBRBs). It is currently underway under the supervision of Prof. Bin Luo of the Australian Institute for Bioengineering and Nanotechnology (AIBN) at the University of Queensland, and Xingchen Yang a third year doctorate student from the Beijing University of Chemical Technology (BUCT).
For an introduction to the ongoing research in the field of zinc bromine flow batteries, refer to AIBN - Zinc–Bromine Rechargeable Batteries - Nano-Micro Letters, a synthesis of a 2023 review.
The battery system studied uses a Daramic seperator, often sprayed with a Nafion polymer solubilised in ethanol, sandwiched in between two functionalised porous carbon felt pads in which flows the anolyte (negative electrolyte) and catholyte (positive electrolyte) on their respective sides. Whilst zinc metal is oxidised to
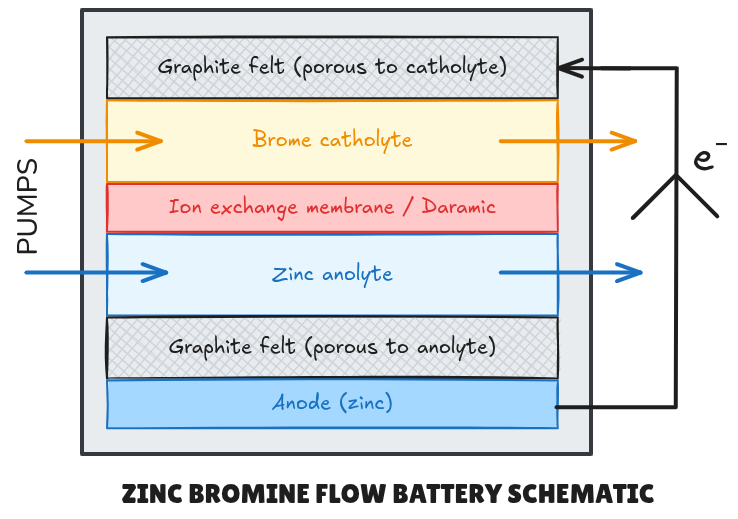
In the context of this system explained here above, thin films of MOF species can be applied in potentially multiple locations via multiple deposition methods. More classically, they can be applied locally on the surface of the zinc anode via spin coating, doctor blade or simply a spray bottle (viscosity allowing). However, dendrite growth often occurs within the graphite felt matrix on the side closest to the separator. A MOF coating could therefore be applied to the zinc side of the separator membrane. This could potentially also reduce complexed or poly bromine diffusion between the half cells.
In order to use MOF in the
The PVDF + NMP mixture used in the previous project for the ESPCI may dissolve the membrane : therefore new MOF plus binder combinations, must be explored. Potential alternatives are listed below :
- Usage of a slurry to avoid excessive membrane dissolution
- Changing the solvent to avoid membrane dissolution (usage of alternative water soluble binders)
- MOF + nafion combination, as this polymer is a proven choice for daramic coatings
In addition, it will be worth investigating the film application from the following methods:
- simple doctored blade application (or slot-die coating)
- usage of a spray bottle
- spin coating
In order to quantify their effects, film homogeneity will be evaluated via optical microscopy and thickness via a micrometer caliper. If possible, SEM imaging could help confirm suspicions established via these macro characterisation methods.
Once completed, the stable binder + MOF combinations can be evaluated on their effect on bromine diffusion across the separator